Have an appreciation for the difference between atomic 
In a lot of very broad, high-level terms, you can kind of view it as being very close to It's roughly equal toĪn atomic mass unit." It's not exactly an atomic mass unit, but, roughly speaking, So, from this, you can say, "Hey, look, if I add a neutron. Looks like it's plusġ.0034 atomic mass units. So, when you look at theĭifference in atomic mass, notice the change is. And this, right over here, is gonna have one more The six protons are what make it carbon, so both of these will have six protons. You might want to note is, what's the difference betweenĬarbon 12 and carbon 13? Carbon 12, this right So, I can write thisĪs approximately 12.01. 
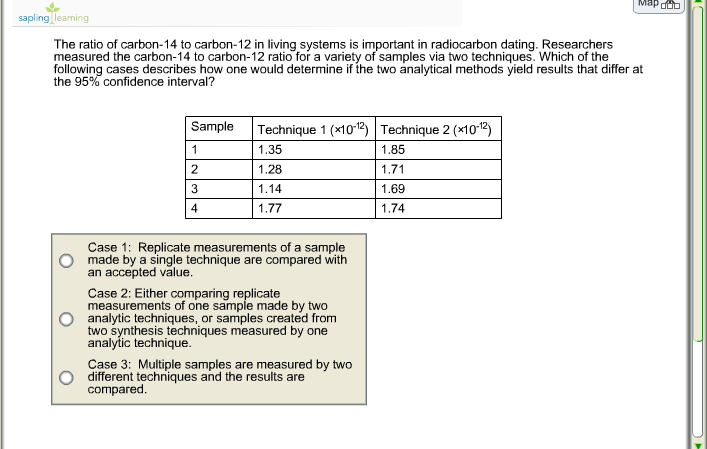
To the hundredths place, is how this atomic weight was gotten. And so, that's all going to be,Īs you can see, 12.01113774, which, if you were to round This multiplication first because it's a calculator knowsĪbout order of operations. So, we are going to have 0.9889 times 12 is equal to 11.8668. So, what does that give us? Let's get our calculator out here. So, what we wanna do is, we could take 98.89%Īnd multiply it by 12. And what do we weight it by? We weight it by how common So, to find this roughly 12.01, we take the weightedĪverage of these two things. Is the weighted average of the atomic masses of the various isotopes of that element. Weight and on atomic mass, we see that the atomic weight Where does that come from? Well, in the video on atomic Periodic table like that? So, atomic weight. The atomic weight number that they'll give you on a Think about, in this video, is how do they come up with So, these numbers that we have here, just as a review, these are atomic mass. And we can experimentally find that its mass is 13.0034 atomic mass units. 1.11% of the carbon on Earth is carbon 13. And we know that, by definition, its mass is exactly 12 atomic mass units. We know that carbon 12 is the most common isotope of carbon on Earth. The rest of it stays, and that is why carbon-13 has a mass of 13.0034 amu. In fact, we only need to convert 0.0046 amu into energy. Now, it seems like carbon-13 should have a mass of 13, because it seems like all of the extra mass from the neutrons and protons turns into binding energy, right? Nope! In this case, we don't need to lose the whole 0.008 amu worth of energy to keep that extra neutron bound in place. If you put the energy back in, then the nucleus would fly apart.Īnyway, back to carbon-13. Because of this energy being released, a carbon-12 nucleus is more stable than six protons and six neutrons are by themselves. So why does C-12 have less mass than the sum of its components? The extra mass is converted to energy (E = mc2), which is released. If you add the masses of six protons and six neutrons, you get 12.09. The actual mass of a proton is about 1.007 amu, and the mass of a neutron is about 1.008 amu. Theoretically, this would mean that each proton and each neutron has a mass of one amu, but this turns out not to be so. MARR is 8 percent/year.It's because of something called binding energy.Ĭarbon-12 has a mass of 12 amu by definition. The expectedĬash flows for each machine are shown below. Technological change in this area, it is realistic to assume that these are one-shot investments. Does this have any advantages or disadvantages over the full survey in part (b) in terms of possible bias? Explain.Įchnology Innovations is planning to purchase one of two chip insertion machines. Only the residents in the sample are to be made aware of the survey through information mailed with their utility bills, and asked to go online to complete the survey. (c) Suppose we select a random sample of utility customers. Discuss any sources of bias that might be contained in the full survey. Residents are to be made aware of the survey through information mailed with their utility bills, and asked to go online to complete the survey. The full survey is to be an online survey. (b) The survey at the Earth Day celebration was intended to be used as a pilot study to make sure the questions were not ambiguous. (a) If the population of intere st is adult residents of the city, discuss any sources of bias that might limit the usefulness in using this survey to draw conclusions about this population. They set up an information booth at the city's Earth Day celebration, and people who visit the booth are offered the opportunity to complete a survey on their knowledge of stormwater pollution. The administrators of the city's Stormwater Program are interested in evaluating the public's knowledge of the causes and effects of stormwater pollution. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
